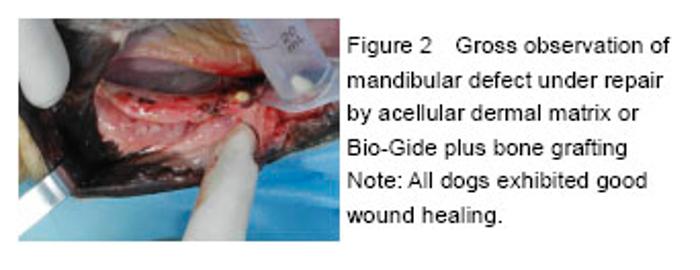
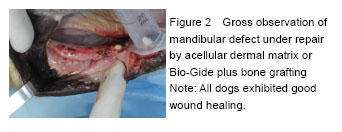
Chinese Journal of Tissue Engineering Research ›› 2016, Vol. 20 ›› Issue (30): 4442-4448.doi: 10.3969/j.issn.2095-4344.2016.30.005
Previous Articles Next Articles
Guided bone regeneration with acellular dermal matrix as a barrier for bone defects
Jia Ren-jie1, Ren Yu-qing1, Xu Hao2, Wang Wei-ying2, Yi Zhong-ping2, Zhao Bao-dong2
- 1 Qingdao University, Qingdao 266000, Shandong Province, China
2 Department of Oral Implantology, Affiliated Hospital of Qingdao University, Qingdao 266100, Shandong Province, China
-
Received:2016-05-11Online:2016-07-15Published:2016-07-15 -
Contact:Zhao Bao-dong, Chief physician, Professor, Department of Oral Implantology, Affiliated Hospital of Qingdao University, Qingdao 266100, Shandong Province, China -
About author:Jia Ren-jie, Studying for master’s degree, Qingdao University, Qingdao 266000, Shandong Province, China Ren Yu-qing, Studying for master’s degree, Qingdao University, Qingdao 266000, Shandong Province, China Jia Ren-jie and Ren Yu-qing contributed equally to this work. -
Supported by:the Natural Science Foundation of Shandong Province, China, No. ZR2010HM036
CLC Number:
Cite this article
Jia Ren-jie, Ren Yu-qing, Xu Hao, Wang Wei-ying, Yi Zhong-ping, Zhao Bao-dong. Guided bone regeneration with acellular dermal matrix as a barrier for bone defects[J]. Chinese Journal of Tissue Engineering Research, 2016, 20(30): 4442-4448.
share this article
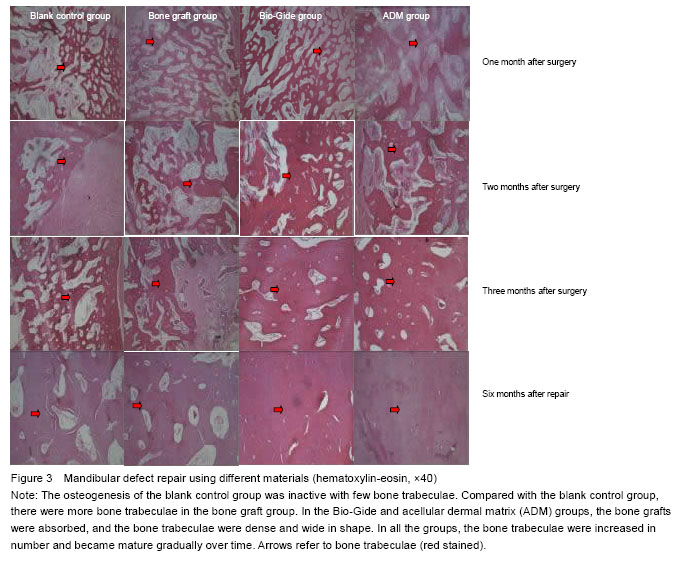
Findings from histological observation No serious inflammatory reaction occurred in all groups during the experiment. One month after the surgery, the osteogenesis of the blank control group was inactive, accompanied by a large amount of fibrous connective tissues occupying the bone defect area. Moreover, the bone trabeculae were in small quantity, which were only found at bottom but not at the center and the top of the defect region. In the bone graft group, it could be seen that a mass amount of fibrous connective tissues were interspersed in the bone implant. Compared with the blank control group, the number of original bone trabeculae was higher and the interval between the trabeculae was relatively wider in the bone graft group. Additionally, the bone trabeculae in the bone graft group were mostly gathered at the bottom, but less at the center and the top. In the ADM and Bio-Gide groups, the coated connective tissues did not stretch obviously; compared with the other two group, the bone trabeculae were larger in shape with a smaller interval, which were distributed evenly in the visual field (Figure 3). After 2-3 months, new bone tissues were gradually formed and became mature (Figure 3). After 6 months, a marked increase in new bone tissues was found in all the groups. In the blank control group, a great number of fibrous connective tissues occupied the defect region, but there were less regenerated and mature bone tissues that were mainly located at the bottom. In the bone graft group, there were more regenerated and mature bone tissues, with the presence of compact bone at bottom, a few fibrous tissues at the central and little bone tissues at the top. The bone trabeculae were interspersed from the top to bottom. In the ADM and Bio-Gide groups, a large amount of mature bone tissues filled in the defect region, accompanied by little connective tissues; and no mature lamellar bone was formed, indicating no obvious regional difference (Figure 3)."
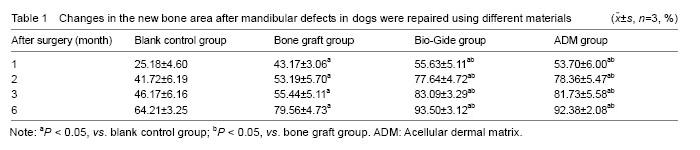
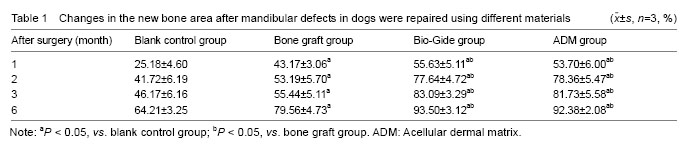
Measurement of new bone area Compared with the blank control group, the percentage of new bone area was increased significantly in the other three groups (P < 0.05), which was increased higher in the Bio-Gide and ADM groups followed by the bone graft group (P < 0.05). Moreover, there was no significant difference between the Bio-Gide and ADM groups (P > 0.05; Table 1)."
| [1] Cafiero C, Annibali S, Gherlone E, et al. Immediate transmucosal implant placement in molar extraction sites: a 12-month prospective multicenter cohort study. Clin Oral Implants Res. 2008;19(5):476-482.
[2] Nikol'ski? VIu. Morphologic analysis of reparative osteogenesis in cases of immediate dental implantation in experiments on rabbits. Stomatologiia (Mosk). 2005; 84(3):8-12.
[3] Taga ML, Granjeiro JM, Cestari TM, et al. Healing of critical-size cranial defects in guinea pigs using a bovine bone-derived resorbable membrane. Int J Oral Maxillofac Implants. 2008;23(3):427-436.
[4] Yu YH, Deng FL. Application and research progress of barrier membrane in GBR Technology. Zhongguo Kouqiang Zhongzhi Xue Zazhi. 2006;11(3):137-140.
[5] Chen HL, Sun Y. Review and Prospect of guided bone regeneration technique in oral and maxillofacial defect. Xinan Guofang Yiyao. 2009;19(7):752-753.
[6] Qiu LX, Wang Y, Lin Y, et al. Application of biological membrane technology guided bone regeneration in the implant denture. Zhonghua Kouqiang Yixue Zazhi. 1998;33(1):58-59.
[7] Camelo M, Nevins ML, Schenk RK, et al. Clinical, radiographic, and histologic evaluation of human periodontal defects treated with Bio-Oss and Bio-Gide. Int J Periodontics Restorative Dent. 1998;18(4): 321-331.
[8] Camelo M, Nevins ML, Lynch SE, et al. Periodontal regeneration with an autogenous bone-Bio-Oss composite graft and a Bio-Gide membrane. Int J Periodontics Restorative Dent. 2001;21(2):109-119.
[9] Du Y, Yu XJ, Lin J. Long-term clinical evaluation of periodontal defects treated with Bio-gide and Bio-oss. Linchuang Kouqiang Yixue Zazhi. 2007;23(11):684-686.
[10] Yu YH, Deng FL, Fu Y. Advances of accelluar dermal matrix application in dentistry. Zhonghua Kouqiang Yixue Zazh. 2006;41(9):575-576.
[11] Hu XL, Lin Y, Wang J, et al. Clinical study of tissue preservation of extraction socket with Bio-Oss collagen and acellular dermal matrix. Zhonghua Kouqaing Yixue Zazhi. 2009;44(9):513-516.
[12] Novaes AB Jr, Souza SL. Acellular dermal matrix graft as a membrane for guided bone regeneration: a case report. Implant Dent. 2001;10(3):192-196.
[13] Wang QF, Liu HW, Zheng QL. Histological changes of acellular dermal matrix used as guided tissue regeneration barrier membrane in vivo. Kouqiang Yixue. 2010;30(9):523-525.
[14] Wang SM. Experimental study of repairing the mandibular bone defect with platelet-rich plasma combined particulate of autogenous cancellous bone. Taiyun: Shanxi Medical University, 2007.
[15] Guo ZM, Rong MD, Zhu AD, et al. Bone metrology of hard tissue containing implant: undecalcified plastic embedding technology. Guangdong Yixue. 2008;29(3): 385-386.
[16] Wang DS. Modified method of making ground section of bone with dental implant. Kouqiang Hemian Xiufu Xue Zazhi. 2006;7(3):169-170.
[17] Yukna RA, Turner DW, Robinson LJ. Variable antigenicity of lyophilized allogeneic and lyophilized xenogeneic skin in guinea pigs. J Periodontal Res. 1977;12(3):197-203.
[18] Livesey SA, Herndon DN, Hollyoak MA, et al. Transplanted acellular allograft dermal matrix. Potential as a template for the reconstruction of viable dermis. Transplantation. 1995;60(1):1-9.
[19] Walter RJ, Matsuda T, Reyes HM, et al. Characterization of acellular dermal matrices (ADMs) prepared by two different methods. Burns. 1998;24(2): 104-113.
[20] Zuo JH, Yang YC, Li WX, et al. Study on the hioperties of allogenic acellular derunal matrix. Linchuang Kouqiang Yixeu Zazhi. 2004;20(5):276-278.
[21] Huang YJ, Zhang JC, Liu SG, et al. Establishment of animal model of peri-implant bone defect. Guangdong Yixue. 2010;31(18):2340-2342.
[22] Feng LF, Wu L. The evaluation methods of dental implant in canine alveolar bone and bone integration. Zhongguo Shiyong Kouqiang Ke Zazhi. 2013;6(8):496-502.
[23] Bao JB, Xie ZG, Zhang Y, et al. Establishment of beagle dogs model for the study of the dental implant. Zhongguo Kouqiang Zhongzhi Xue Zazhi. 2012;17(2):58-61.
[24] Li ZR, Xu S, Zhou WJ, et al. Estabilishment of an experimental animal model of immediately peri-imllant bone defects on Beagle dogs. Zhongguo Kouqiang Zhongzhi Xue Zazhi. 2012;17(1):14-17.
[25] Wikesjö UM, Sorensen RG, Kinoshita A, et al. RhBMP-2/alphaBSM induces significant vertical alveolar ridge augmentation and dental implant osseointegration. Clin Implant Dent Relat Res. 2002; 4(4):174-182.
[26] Wikesjö UM, Qahash M, Thomson RC, et al. rhBMP-2 significantly enhances guided bone regeneration. Clin Oral Implants Res. 2004 ;15(2):194-204.
[27] Tatakis DN, Koh A, Jin L, et al. Peri-implant bone regeneration using recombinant human bone morphogenetic protein-2 in a canine model: a dose-response study. J Periodontal Res. 2002;37(2):93-100.
[28] Song YP, Xu ST, Yang SJ, et al. The contrastive research on the osteogensis effects of recombinanr human bone morphogenetiv protein-2, platelet-rich fibrin and autologous bone compositeding with coralline hydroxyapatite respectively. Zhongguo Kouqiang Zhongzhi Xue Zazhi. 2014,19(4):155-159.
[29] Zhang Y, Liu XN, Li TW, et al. Osteogenic efficiency of antigen-extracted xenogeneic cancellous bone. Zhongguo Bingli Shengli Zazhi. 2014;30(8):1467-1471.
[30] Xu ST, Zhou L, Yang XY, et al. Clinical study of guided bone regeneration in peri-implant bone defects with combining titanium membrane and collagen membrane. Kouqiang Yixue Yanjiu. 2009;25(5):619-621. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wang Yujiao, Liu Dan, Sun Song, Sun Yong. Biphasic calcium phosphate loaded with advanced platelet rich fibrin can promote the activity of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 504-509. |
| [5] | Liu Jiangfeng. Nano-hydroxyapatite/polyamide 66 composite filling combined with locking plate in the treatment of fibrous dysplasia of femoral bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 542-547. |
| [6] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [7] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [8] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [9] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [10] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [11] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [12] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [13] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [14] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [15] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||